
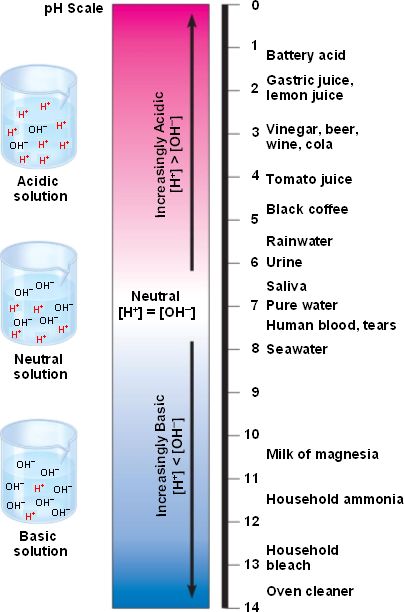
 The concentration of hydrogen ions (H+, or protons) in aqueous solution
is measured in pH units.
The concentration of hydrogen ions (H+, or protons) in aqueous solution
is measured in pH units.
The H+ concentration at 25°C ranges from 1 Molar (100, or pH=0) to 10-14 M (pH=14).
These concentrations are plotted on a negative logarithmic scale: each unit change in pH is a 10-fold change in H+ ion concentration.
An acid has a high concentration of H+ ions (low pH).
A base has a low H+ concentration (high pH).